Medford (541) 770-2020
Grants Pass (541) 956-2020
Email Us
Patient Portal



Welcome to
Oregon Retina Center
As a leading eye care center in Southern Oregon, our practice is built on its commitment to excellence. We specialize in medical and surgical eye care for diabetes, macular degeneration, and other retina-related eye services. We provide advanced treatments using state-of-the-art technology at our Medford and Grants Pass offices.
Expert Eye Care
Diabetes Eye Care
Our Mission
Our mission is to provide you with the highest quality medical & surgical eye & retina care in a caring and compassionate manner. Advanced therapies complement our state of the art technology in tailoring treatments that improve and preserve vision.
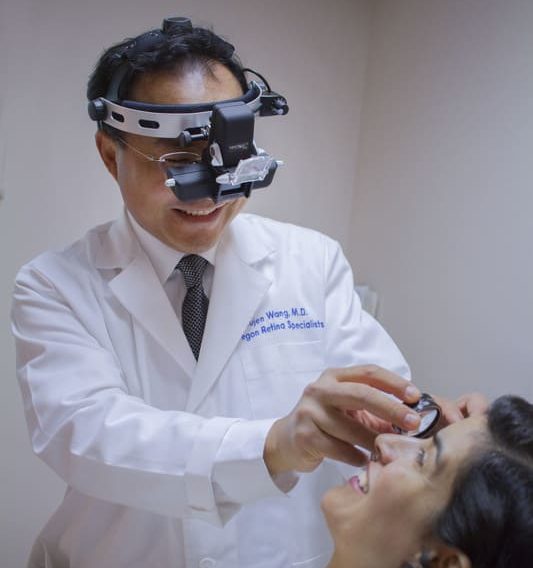
If you’re interested in learning more about our specialized retina care, call our Medford office at (541) 770-2020 or our Grants Pass office at (541) 956-2020. We’re here to help!
Oregon Retina Center
Medford
1518 E Barnett Rd
Medford, OR 97504
(541) 770-2020
Write us a review
Grants Pass
1867 Williams Hwy
Grants Pass, OR 97527
(541) 956-2020
Write us a review
Contact
Monday - Friday: 8:00 AM - 5:00 PM
info@oregonretina.com
Proudly Serving Southern Oregon Communities
Jackson, Josephine, The Applegates, Ashland, Butte Falls, Central Point, Cave Junction, Eagle Point, Grants Pass, Jacksonville, Lake Creek, Medford, Merlin, Murphy, Phoenix, Prospect, Provolt, Rogue River, Ruch, Sams Valley, Shady Cove, Sunny Valley, Talent, Trail, White City, Williams, Wolf Creek

